|
|
Post by 1dave on Jan 3, 2023 12:25:34 GMT -5
I'm coppying this post from elsewhere as I have long puzzled about this fact. mohs copper IS added to Sterling silver coins to make them harder and not wear away so fast. I find it interesting that the " Precocious Metals" all have 11 valance electrons. 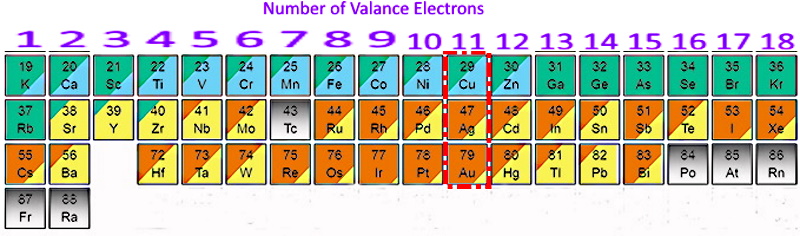 |
|
|
|
Post by 1dave on Jan 3, 2023 12:34:50 GMT -5
Question: What do the 11 electrons do to make these three elements so different from all others?
|
|
|
|
Post by jasoninsd on Jan 3, 2023 17:42:01 GMT -5
That's actually a very fascinating observation Dave! I have ZERO education or knowledge to harbor a sound reason. I can say eleven is the first double-digit prime number though. Oh...and I was commonly referred to as a "precocious" child...never a "precious" child though!  |
|
|
|
Post by 1dave on Jan 3, 2023 18:06:10 GMT -5
That's actually a very fascinating observation Dave! I have ZERO education or knowledge to harbor a sound reason. I can say eleven is the first double-digit prime number though. Oh...and I was commonly referred to as a "precocious" child...never a "precious" child though!  I hope you aren't the only thinker in this group. |
|
|
|
Post by jasoninsd on Jan 3, 2023 18:51:18 GMT -5
That's actually a very fascinating observation Dave! I have ZERO education or knowledge to harbor a sound reason. I can say eleven is the first double-digit prime number though. Oh...and I was commonly referred to as a "precocious" child...never a "precious" child though!  I hope you aren't the only thinker in this group. God help us all if I am! LOL |
|
|
|
Post by hummingbirdstones on Jan 3, 2023 18:59:30 GMT -5
Just call me clueless.  I had to look up what a valance electron was. Per Wiki: In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair forms with both atoms in the bond each contributing one valence electron. The presence of valence electrons can determine the element's chemical properties, such as its valence—whether it may bond with other elements and, if so, how readily and with how many. In this way, a given element's reactivity is highly dependent upon its electronic configuration. For a main-group element, a valence electron can exist only in the outermost electron shell; for a transition metal, a valence electron can also be in an inner shell. An atom with a closed shell of valence electrons tends to be chemically inert. Way too much stuff in this one paragraph I'd have to look up just to understand what it said! |
|
|
|
Post by jasoninsd on Jan 3, 2023 19:02:12 GMT -5
Just call me clueless.  I had to look up what a valance electron was. Per Wiki: In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair forms with both atoms in the bond each contributing one valence electron. The presence of valence electrons can determine the element's chemical properties, such as its valence—whether it may bond with other elements and, if so, how readily and with how many. In this way, a given element's reactivity is highly dependent upon its electronic configuration. For a main-group element, a valence electron can exist only in the outermost electron shell; for a transition metal, a valence electron can also be in an inner shell. An atom with a closed shell of valence electrons tends to be chemically inert. Way too much stuff in this one paragraph I'd have to look up just to understand what it said!  (You're not the only one Robin!!  ) |
|
|
|
Post by mohs on Jan 3, 2023 19:09:27 GMT -5
|
|
|
|
Post by hummingbirdstones on Jan 3, 2023 19:16:00 GMT -5
Just call me clueless.  I had to look up what a valance electron was. Per Wiki: In chemistry and physics, a valence electron is an electron in the outer shell associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed. In a single covalent bond, a shared pair forms with both atoms in the bond each contributing one valence electron. The presence of valence electrons can determine the element's chemical properties, such as its valence—whether it may bond with other elements and, if so, how readily and with how many. In this way, a given element's reactivity is highly dependent upon its electronic configuration. For a main-group element, a valence electron can exist only in the outermost electron shell; for a transition metal, a valence electron can also be in an inner shell. An atom with a closed shell of valence electrons tends to be chemically inert. Way too much stuff in this one paragraph I'd have to look up just to understand what it said!  (You're not the only one Robin!!  ) Chemistry was never my forte, Jason. Just ask my high school chemistry teacher!  |
|
|
|
Post by vegasjames on Jan 3, 2023 19:24:56 GMT -5
Question: What do the 11 electrons do to make these three elements so different from all others? You cannot simply look at the valence electrons as to what makes all the metals different. Different atoms have varying numbers of electrons not only in the outer shell, but also the other shells. As well as varying numbers of protons and neutrons.
It is like all humans share 99.9% of the same DNA yet look at how different we all are from just that little difference in the last 0.1%.
|
|
|
|
Post by Son Of Beach on Jan 3, 2023 20:23:05 GMT -5
Chemistry fight, go! mohs I'm still not convinced you didn't see a cinnamon bun at the counter. |
|
|
|
Post by mohs on Jan 3, 2023 20:33:23 GMT -5
mohs I'm still not convinced you didn't see a cinnamon bun at the counter. haha Chad doesn't take much to put m  hs into glazed over trance mosstly o I just wanted say I don't always think butte when I do I ponder deeply into 1dave threads  |
|
|
|
Post by 1dave on Jan 3, 2023 21:09:55 GMT -5
Question: What do the 11 electrons do to make these three elements so different from all others? You cannot simply look at the valence electrons as to what makes all the metals different. Different atoms have varying numbers of electrons not only in the outer shell, but also the other shells. As well as varying numbers of protons and neutrons.
It is like all humans share 99.9% of the same DNA yet look at how different we all are from just that little difference in the last 0.1%.
Hi Vegas, I've always known that you are one of the thinkers at RTH. We Know with 11 outer ring electrons there are 2 in the middle, 8 in the next, and 1 electron left in the outer ring giving a normal valence of ONE. For gold that tends to keep the metal fairly pure. Does it help silver and copper the same way? |
|
dillonf
fully equipped rock polisher
   Hounding and tumbling
Hounding and tumbling
Member since February 2022
Posts: 1,577
|
Post by dillonf on Jan 3, 2023 22:00:34 GMT -5
You cannot simply look at the valence electrons as to what makes all the metals different. Different atoms have varying numbers of electrons not only in the outer shell, but also the other shells. As well as varying numbers of protons and neutrons.
It is like all humans share 99.9% of the same DNA yet look at how different we all are from just that little difference in the last 0.1%.
Hi Vegas, I've always known that you are one of the thinkers at RTH. We Know with 11 outer ring electrons there are 2 in the middle, 8 in the next, and 1 electron left in the outer ring giving a normal valence of ONE. For gold that tends to keep the metal fairly pure. Does it help silver and copper the same way? Yes, elements with the same number of valence electrons and in the same group tend to have similar reactivity. |
|
|
|
Post by vegasjames on Jan 4, 2023 0:20:03 GMT -5
You cannot simply look at the valence electrons as to what makes all the metals different. Different atoms have varying numbers of electrons not only in the outer shell, but also the other shells. As well as varying numbers of protons and neutrons.
It is like all humans share 99.9% of the same DNA yet look at how different we all are from just that little difference in the last 0.1%.
Hi Vegas, I've always known that you are one of the thinkers at RTH. We Know with 11 outer ring electrons there are 2 in the middle, 8 in the next, and 1 electron left in the outer ring giving a normal valence of ONE. For gold that tends to keep the metal fairly pure. Does it help silver and copper the same way?
So here is something for you to really ponder. Atoms are not solid. They are primarily empty space with electrons, protons and neutrons that are not really solid. Therefore, we are not really solid, nor are things such as walls. So why do we appear as a solid as does the wall even though nothing about the atoms that make up us or walls are solid? Why can't wee simply pass though the walls that are mainly empty space just like us? The answer is actually quite simple.
|
|
|
|
Post by mohs on Jan 4, 2023 1:48:28 GMT -5
Here an imponderable !
I have put my hand & arm and through a brick wall
Literally
I looked at my hand sticking through
the other side of said wall
Honestly
It was on chemical substance
sure the valence of this substance
caused some warping of the empedoclian
two major forces
leave ya to ponder what those 2 major forces be
Along with the 4 elements
Which I believe constitute solid reality
Mostly
|
|
|
|
Post by vegasjames on Jan 4, 2023 2:24:43 GMT -5
Here an imponderable ! I have put my hand & arm and through a brick wall Literally I looked at my hand sticking through the other side of said wall Honestly It was on chemical substance sure the valence of this substance caused some warping of the empedoclian two major forces leave ya to ponder what those 2 major forces be Along with the 4 elements Which I believe constitute solid reality Mostly Technically through a hole in the brick wall. |
|
|
|
Post by rockpickerforever on Jan 4, 2023 5:37:39 GMT -5
 (You're not the only one Robin!!  ) Chemistry was never my forte, Jason. Just ask my high school chemistry teacher!  Is the science considered chemistry, molecular physics, or something else? I only know I've sucked at it since junior college. Don't feel bad, Robin. I think it takes a certain kind of person to Grok that stuff! |
|
|
|
Post by mohs on Jan 4, 2023 9:37:03 GMT -5
|
|
|
|
Post by 1dave on Jan 4, 2023 9:44:05 GMT -5
From vegasjamesExceptionsFrom the table above, you must have noticed that in the electronic configuration of chromium (Cr) and copper (Cu), there is only 1 electron placed in the 4s (outer shell) orbital after which filling starts in the (3d) inner subshell. The transition metal elements in groups 6 and 11 are thus called exceptions. This is because a half-filled 3d5 or a completely filled 3d10 subshell is more stable than the partially filled d4 or d9 respectively. However, it does not effect the total valence electrons present in each case. exception in electron configuration Variable valencies of transition metal elements We discussed at the beginning of this article that transition metal elements can involve their inner shell valence electrons in addition to the outer electrons in chemical bonding. Therefore, transition metal elements exhibit variable valencies. They can form more than one type of ion.For example, Iron (Fe) has a total of 8 valence electrons that it can use for chemical bonding. It most commonly loses its two 4s electrons from the outermost shell to form Fe2+ (ferrous) ions. But, it can also lose three valence electrons (two electrons from 4s and 1 electron from the inner 3d orbital to from Fe3+ (ferric) ions. Valence electrons of transition metals (d-blocks) - Why don’t transition metals use all their valence electrons in bonding? Transition metals form positively charged ions by the loss of their valence electrons, prior to chemical bonding. It is due to the force of attraction between the positively charged nucleus and the negative electrons that the transition metal atom cannot lose all of its valence electrons at once for bonding. What is the difference between the valence electrons and the valency of transition metals? The valence electrons are the total electrons available for chemical bonding while valency refers to the combining power of the transition metal element with other elements. Summary
Transition metals are situated in the d and f blocks of the Periodic Table.
In addition to their outer-shell electrons, the transition metal elements can involve their inner-shell electrons in chemical bonding.
The inner d-subshell electrons of transition metals are also called valence electrons.
So, the valence electrons of a transition metal can be determined using its electronic configuration, by taking the sum of its ns and (n-1)d electrons.
First-row transition metals have a similar electronic configuration that ends in 3d1-10 4s2.
24Cr and 29Cu are exceptions with a half-filled 4s1 atomic orbital. |
|