|
|
Post by vegasjames on Jan 10, 2017 17:08:14 GMT -5
My point still stands. Same chemistry, different structure still makes a completely different thing. Let me put it this way. If I write the chemical formula C 6H 12O 6 am I referring to glucose, fructose, mannose, tagatose or galactose? All have the same exact formula C 6H 12O 6 as do some other sugars yet they all have different chemical structures making them all different compounds with different properties. But all made of the same exact stuff. Yes, and cows are made up of the same chemicals as humans but that does not mean cows are human. |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on Jan 10, 2017 17:15:03 GMT -5
The question was this: Is obsidian the same chemistry as granite. Yes? No? Maybe so? It appears that it indeed is or at least can be. No. Obsidian ain't granite nor the reverse. But chemistry is similar or same. That is all. Sorry you got sucked into the morass of something else. I hope you are well. vegasjames1dave |
|
|
|
Post by vegasjames on Jan 10, 2017 20:31:07 GMT -5
The question was this: Is obsidian the same chemistry as granite. Yes? No? Maybe so? It appears that it indeed is or at least can be. No. Obsidian ain't granite nor the reverse. But chemistry is similar or same. That is all. Sorry you got sucked into the morass of something else. I hope you are well. vegasjames 1dave Still overlooking my point. Just because two or more things have the same atoms as each other this does not make them the same chemically. Part of chemistry is also atomic arrangement. This is why as I pointed out glucose, fructose, mannose, tagatose or galactose and some other sugars all have the same exact atoms of numbers of those atoms yet the are still completely different compounds as their atomic arrangement, and thus their chemistry, is different. So to claim obsidian is granite just based on having the same atoms is ridiculous. And yes, things going good. Just got back from the turquoise mine again the other day and did pretty good. And Jax had a lot of fun since we have been able to get out there in a while. |
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 11, 2017 15:26:39 GMT -5
Yes, obsidian can have a granitic composition. Normally it is basaltic. NB the terms 'Granitic (continental / SiAl)' composition and 'Basaltic (oceanic / SiMa)', are the two root-families of igneous geochemistry. This is an assignment of the melt species at a family level. Recall the basic paradigm of classification involves a level of generalisation. As per usual, the scale of the claim was macroscopic where the scale of examination here tends to the microscopic. Not knowing how much the source of the statement knows, it is tempting to say they were wrong. Of course there are often exceptions. vegasjames suggestion for inequality appears to stem from the point of view that it can't be the same due to the different granularity. Note that for obsidian, we're talking about glass. Glass is by definition out of equilibrium (and instead, equalibrated with the conditions at the time of formation. Even the term cryptocrystalline can be erroneous since the melt is quenched. Strictly speaking it often remains a fluid. In the UK there are very old church (etc) windows that have flowed so the bottom is thicker than at the top. However it still hasn't crystallised. The example case of a different glass - toffee - should illustrate the compositional argument well. Cryptocrystalline fudge, it could be said is completely different to toffee. In fact, leave it too long and it crystallises out; some call that fudge too. At a microscopic level, the composition of the melt has changed certainly, but boil it all up again and toffee, it can be once again. Coincidentally I found this!Composition varies all through the pluton with a 360 degree basis of variation; think to the occurrence of inclusions in crystals; reaction with the host rock, xenoliths, heat / cooling and pressure / release. It could be said that the first eruptive material to blow out of a volcano is different to that in the pipes behind it and the pluton otherwise it wouldn't have blown out first. It may be the petroleum from the crude, but generally not so well fragmented, noting the timing of magmatic ascent can be very rapid. However, as soon as the blow-out occurs, a huge change is initiated due to the change in pressure and particularly the hydrous phases and viscosity. Donald Dingwell ran a course on Volcanic Eruptions on Coursera (it's not there now, but may be again in future if you're interested - but *warning* ... the geochemistry and for me, at least, the visco-elastic relationship of structural relaxation was a head-scrambler). The essence of his delivery can be found here . |
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 11, 2017 15:33:30 GMT -5
blah, blah... The example case of a different glass - toffee - should illustrate the compositional argument well. Cryptocrystalline fudge, it could be said is completely different to toffee. In fact, leave it too long and it crystallises out; some call that fudge too. At a microscopic level, the composition of the melt has changed certainly, but boil it all up again and toffee, it can be once again. Coincidentally I found this!blah, blah... 'Cept they're wrong ... 'This is like a volcanic igneous rock that cooled quickly above the earth’s surface, like obsidian.' No, the metaphorical equivalent of that would be hard, clear, glassy toffee! |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on Jan 12, 2017 13:07:08 GMT -5
metalsmithChas that was amazing! Clicking links soon. And you have me an idea to test a product. ETA: re toffee analogy Obsidian isn't often clear. Therefore, I suggest toffee must not necessarily be either! Haha! Must learn toffee better to understand plutonics!!
|
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 12, 2017 13:48:59 GMT -5
metalsmithChas that was amazing! Clicking links soon. And you have me an idea to test a product. ETA: re toffee analogy Obsidian isn't often clear. Therefore, I suggest toffee must not necessarily be either! Haha! Must learn toffee better to understand plutonics!! No problem.  Not all toffee is clear. Not all glass is transparent; I found a paper where the crystal content of obsidian was stated as ranging between 5-15% for one locality. Glass is more of a phase description, seemingly not fitting with any of solid liquid or gas: leads me to this. More links! Read Glass through Liquid Crystal States - it's all relevant. For the geochemical composition, search on what constitutes rhyolite - it's the same stuff (generally!) Enjoy! |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on Jan 12, 2017 14:58:41 GMT -5
|
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 12, 2017 17:34:09 GMT -5
|
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 13, 2017 3:12:10 GMT -5
@shotgunner Ahh... you got carried away with states of matter, I see! In a quantum spin you may be. Dangerous, it is to play with neutron-degenerate matter... Neuron degenerating it can be! You must unlearn before you can learn.   Superfluid with zero viscosity (or infinite fluidity; i.e., flowing without friction), was discovered in 1937 for helium, which forms a superfluid below the lambda temperature of 2.17 K. In this state it will attempt to "climb" out of its container. It also has infinite thermal conductivity so that no temperature gradient can form in a superfluid. Placing a superfluid in a spinning container will result in quantized vortices. A quantum vortex “carries” quantized angular momentum, thus allowing the superfluid to rotate; in a superconductor, the vortex carries quantized magnetic flux. Scotty, we need more power! |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,560
|
Post by jamesp on Jan 13, 2017 9:35:38 GMT -5
Interesting conversation. Steel does phase changes when heated. Water too. Phases changes complicated. SiO2 a very common mineral made simply of silicon and oxygen with complicated and varied mechanical properties. Past that you guys can figure it out. And light. very common, uh, particle ?, wave ? Who knows ? Man may claim to know what light is but in fact he has no idea. Phase diagrams, sure to give headaches. Every mechanical engineer has to become versed in complicated <7% carbon steel phase diagrams. Something of nightmares. Drank my memory free of that study ASAP. Maybe this phase diagram works for the quartz conversation: Crystal phase diagrams[edit] Polymorphic and polyamorphic substances have multiple crystal or amorphous phases, which can be graphed in a similar fashion to solid, liquid, and gas phases. 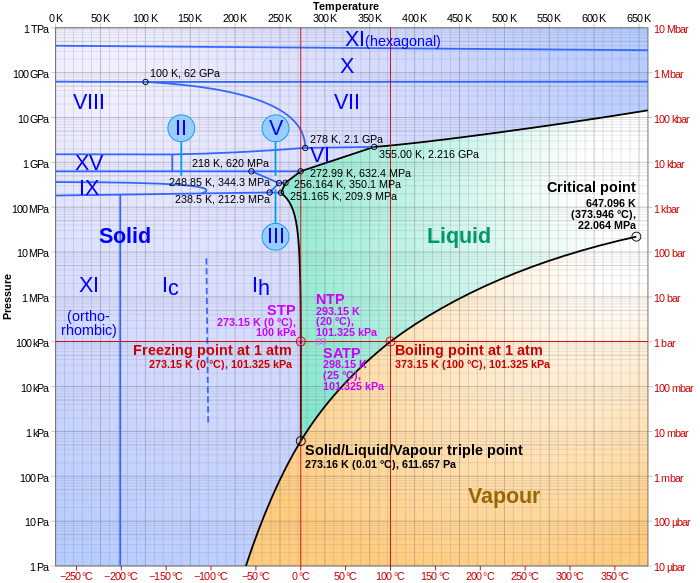 |
|
|
|
Post by Garage Rocker on Jan 13, 2017 10:35:10 GMT -5
No thanks, jamesp. I'll have none of that.^ |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,560
|
Post by jamesp on Jan 13, 2017 10:48:49 GMT -5
No thanks, jamesp. I'll have none of that.^ On that subject we are in the exact same boat. No mas |
|
|
|
Post by 1dave on Jan 13, 2017 10:51:09 GMT -5
Interesting conversation. Steel does phase changes when heated. Water too. Phases changes complicated. SiO2 a very common mineral made simply of silicon and oxygen with complicated and varied mechanical properties. Past that you guys can figure it out. And light. very common, uh, particle ?, wave ? Who knows ? Man may claim to know what light is but in fact he has no idea. Phase diagrams, sure to give headaches. Every mechanical engineer has to become versed in complicated <7% carbon steel phase diagrams. Something of nightmares. Drank my memory free of that study ASAP. Maybe this phase diagram works for the quartz conversation: Crystal phase diagrams[edit] Polymorphic and polyamorphic substances have multiple crystal or amorphous phases, which can be graphed in a similar fashion to solid, liquid, and gas phases.  YES! H 2O and SiO 2 very much alike! |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on Jan 13, 2017 11:02:55 GMT -5
Interesting conversation. Steel does phase changes when heated. Water too. Phases changes complicated. SiO2 a very common mineral made simply of silicon and oxygen with complicated and varied mechanical properties. Past that you guys can figure it out. And light. very common, uh, particle ?, wave ? Who knows ? Man may claim to know what light is but in fact he has no idea. Phase diagrams, sure to give headaches. Every mechanical engineer has to become versed in complicated <7% carbon steel phase diagrams. Something of nightmares. Drank my memory free of that study ASAP. Maybe this phase diagram works for the quartz conversation: Crystal phase diagrams[edit] Polymorphic and polyamorphic substances have multiple crystal or amorphous phases, which can be graphed in a similar fashion to solid, liquid, and gas phases.  YES! H 2O and SiO 2 very much alike! And thusly, one may imagine and, if proper knowledge is present, calculate a world where temps and pressures are such that the fluid that life forms in is silicon dioxide and the blood uses what chemistry to transport chemical energy to power life. DNA analogy as well. Species evolved on that planet and ours could never co-exist. |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,560
|
Post by jamesp on Jan 13, 2017 11:27:24 GMT -5
Way beyond my pea brain Scott S. I can't even get past SiO2 lol.
|
|
|
|
Post by 1dave on Jan 13, 2017 12:19:22 GMT -5
  Crystobalite region is where Spheroids > Thundereggs form. |
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 13, 2017 14:06:24 GMT -5
More obsidian info, rocks and knapping fun here |
|
metalsmith
Cave Dweller 
Member since October 2012
Posts: 1,537
|
Post by metalsmith on Jan 14, 2017 3:13:10 GMT -5
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,560
|
Post by jamesp on Jan 14, 2017 6:29:45 GMT -5
More phase diagrams, run for the hills ! Things of nightmares I tell you. amazing stuff metalsmith. I once bought a scrap load of 1.5" angle iron. Like 40 sticks at 10 feet long. Lovely used material. Some how some way it had been heated to a point it had been hardened to the point a drill bit would not penetrate it. I had welded a trailer frame out of it. Only to find I could not do a lot of needed drilling to hold wood decking down. Phase changed. Trailer frame still sitting in bone yard. Useless. Polyethylene can be amalgamated using a hot air gun. Once it cools the bends put into it stay put. Not sure if that falls in the semi-molten state or phase change category. |
|