unclesoska
freely admits to licking rocks
   All those jade boulders tossed in search of gold!
All those jade boulders tossed in search of gold!
Member since February 2011
Posts: 934
|
Post by unclesoska on May 11, 2017 9:55:48 GMT -5
Limonite hardness runs 4.0-5.5, too soft for these cubes. Thanks for playing!
|
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on May 11, 2017 13:17:54 GMT -5
I also thought limonite or hematite after pyrite pseudomorphs. Some pseudomorphs can be mixes of minerals, too. Mohs 6-7 or greater with red streak doesn't leave a lot of possibilities. Something like piemontite might fit, except I've never seen it in cubes (mostly needles or grains in chalcedony or with quartz)
|
|
|
|
Post by rockpickerforever on May 11, 2017 15:06:44 GMT -5
Don't spend a bunch on a quality halite specimen if it is humid where you live. No humidity here. But I'm not a true specimen collection I would coat it with clear coat or something to protect it. Have had a hanksite specimen for a number of years. It definitely sweats. (Hexagonal crystal, BTW.)
Recently picked up a halite specimen at the swap meet for a couple bucks (guy was trying to sell it as fluorite, pffffft). 


Staying dry so far!
A few pics of a pretty nice pyrite almost-cube I have




And a piece of galena

Makes me think of Borg cube, lol. 


Limonite hardness runs 4.0-5.5, too soft for these cubes. Thanks for playing!
One of the bigger limonite cubes I have



Put a spring on it, and it looks just like one of the brushes in the generator on my old Corvair.
That's all I got for now.
|
|
|
|
Post by captbob on May 11, 2017 16:19:54 GMT -5
You sure that's Halite Jean? Did you lick it?
Halite specimens in Florida "melt" (lose their sharp edges) due to the humidity. I have a couple in a display case that haven't fared toooo badly, but there is a light on in there 24/7 which may help keep the humidity lower.
Amusing seeing high quality (high dollar!) pieces at rock shows down here. Doubt the sellers warn potential buyers.
Kinda like people that sell pyrite suns. <- They do not last if more than a couple are stored together.
|
|
|
|
Post by rockpickerforever on May 11, 2017 16:37:54 GMT -5
You sure that's Halite Jean? Did you lick it? Halite specimens in Florida "melt" (lose their sharp edges) due to the humidity. I have a couple in a display case that haven't fared toooo badly, but there is a light on in there 24/7 which may help keep the humidity lower. Amusing seeing high quality (high dollar!) pieces at rock shows down here. Doubt the sellers warn potential buyers. Kinda like people that sell pyrite suns. <- They do not last if more than a couple are stored together. Almost hate to admit it, but yes, I licked it, lol. Salty! I did brush it off, but suspect the light reddish color may be dust. Not much else you can do with it, can't wash it in water! Like Scott said, it is dry here. I probably won't take any precautionary measures to keep it from melting. Maybe the dust was protecting it? The hanksite, on the other hand has experienced some melting. The person we got if from at a show said to apply a thin coating of mineral oil to keep that from happening. Not a big concern. It probably has a pretty good layer of dust on it by now, lol. |
|
unclesoska
freely admits to licking rocks
   All those jade boulders tossed in search of gold!
All those jade boulders tossed in search of gold!
Member since February 2011
Posts: 934
|
Post by unclesoska on May 11, 2017 16:59:56 GMT -5
Jean, your limonite sure looks like what I have. Would it be possible for you to test the hardness on yours?
|
|
|
|
Post by beefjello on May 11, 2017 18:22:25 GMT -5
So I'm reading this thread thinking "hey I got a pyrite cube.. hey I got a galena cube..". I go and dig them out and find..

okay maybe my pyrite is a cube.. but it's buried in matrix.

okay maybe my galena piece isn't exactly a cube...

the other side with all the druzy crystal things looks like a Borg ship blowing up tho, right rockpickerforever ? 
Sorry I'm not really adding anything to this thread. 
|
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on May 11, 2017 18:43:14 GMT -5
Jeans piece is halite. The pink is from algae in the water that is precipitated from.
|
|
|
|
Post by captbob on May 11, 2017 18:54:15 GMT -5
Yes, mine are pink as well. My question(ing) was in that the cubes are so smooth. Mine are more like this - picture taken from eBay listing.  |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on May 11, 2017 20:09:56 GMT -5
Awesome piece. I love the hollow cubes with edges and vertices. ( jeannie taught me that vocabulary). I saw a cabinet specimen that was all hollow cubes like that. A foot long and high! She and I will in all likelihood hit the Searles Lake event in October. Get our own. |
|
|
|
Post by rockpickerforever on May 11, 2017 21:15:02 GMT -5
Jeans piece is halite. The pink is from algae in the water that is precipitated from. Algae, huh? I never thought of that. Thanks, Scott! |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on May 11, 2017 21:51:11 GMT -5
This is a fun thread!
|
|
gemfeller
Cave Dweller 
Member since June 2011
Posts: 4,024 
|
Post by gemfeller on May 11, 2017 22:22:32 GMT -5
So are spinels. I've always been nuts about this ring featuring natural red spinel octahedrons (same crystal habit as many diamonds).  |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on May 11, 2017 22:40:07 GMT -5
So are spinels. I've always been nuts about this ring featuring natural red spinel octahedrons (same crystal habit as many diamonds).  Cubes have six sides. Are spinel also cubic |
|
|
|
Post by rockpickerforever on May 11, 2017 23:31:59 GMT -5
Jean, your limonite sure looks like what I have. Would it be possible for you to test the hardness on yours? I thought mine looked an awful lot like yours. Someone in our rock club ID'ed mine for me, so I assume hardness-wise, it is what you mentioned earlier, 4.0-5.5. How did you test the hardness of your specimens? |
|
gemfeller
Cave Dweller 
Member since June 2011
Posts: 4,024 
|
Post by gemfeller on May 11, 2017 23:46:22 GMT -5
So are spinels. I've always been nuts about this ring featuring natural red spinel octahedrons (same crystal habit as many diamonds).  Cubes have six sides. Are spinel also cubic Yes. Cubic and isometric are synonyms -- look at the chart I posted upthread. Once again, a cubic crystal is defined by having 3 axes of equal length that intersect at 90 degrees. See the chart image of the octahedron (shape of the spinels shown in the image) inside an imaginary cube. Both are cubic/isometric. Many diamond crystals, also cubic, are octahedrons though crystal habits may vary, i.e., not all diamond crystals are octahedrons but they're all cubic. It can get a little too complicated to explain in a few sentences. The image below shows the simplified geometry. 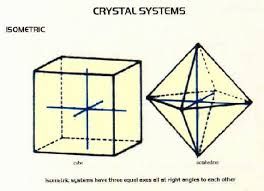 |
|
gemfeller
Cave Dweller 
Member since June 2011
Posts: 4,024 
|
Post by gemfeller on May 11, 2017 23:58:13 GMT -5
Garnets are Isometric Spinel is Cubic I am totally.......!!! Both are cubic. Cubic/isometric mean the same thing in crystallography. But cubic mineral habits can vary. To steal a quote from Wiki: "Garnets are most often found in the dodecahedral crystal habit, but are also commonly found in the trapezohedron habit. (Note: the word "trapezohedron" as used here and in most mineral texts refers to the shape called a Deltoidal icositetrahedron in solid geometry.) They crystallize in the cubic system, having three axes that are all of equal length and perpendicular to each other." Forget all the big words and just remember that garnet, spinel and diamond are the three main cubic gem minerals. There are others of lesser importance. |
|
unclesoska
freely admits to licking rocks
   All those jade boulders tossed in search of gold!
All those jade boulders tossed in search of gold!
Member since February 2011
Posts: 934
|
Post by unclesoska on May 12, 2017 0:31:53 GMT -5
Jean, your limonite sure looks like what I have. Would it be possible for you to test the hardness on yours? I thought mine looked an awful lot like yours. Someone in our rock club ID'ed mine for me, so I assume hardness-wise, it is what you mentioned earlier, 4.0-5.5. How did you test the hardness of your specimens? I have a set of hardness points. You could see if a piece of quartz scratches it. |
|
Deleted
Deleted Member
Member since January 1970
Posts: 0
|
Post by Deleted on May 12, 2017 15:36:34 GMT -5
For the purpose of my simple minded love, a cube is a 3d square and an octahedron doesn't gain my love.
So, only three?
Magnetite (and its iron replacements)
Pyrite
And
Halite
Thats it? What a boring collection. Lol
|
|
gemfeller
Cave Dweller 
Member since June 2011
Posts: 4,024 
|
Post by gemfeller on May 12, 2017 16:00:18 GMT -5
For the purpose of my simple minded love, a cube is a 3d square and an octahedron doesn't gain my love. So, only three? Magnetite (and its iron replacements) Pyrite And Halite Thats it? What a boring collection. Lol See the link to the Mindat list of cubic minerals I posted upthread. I think you'll be more than impressed at the number of specimens you'll need to complete your collection. However many of them will not have the 3d square habit you love. |
|