The Transition Metals - Copper, Silver, and Gold.
www.angelo.edu/faculty/kboudrea/periodic/trans_transition.htmGroup 1B (11)
The Group 1B elements (Group 11 in the IUPAC designation) have the electron configuration (n-1)d10 ns1, instead of the expected (n-1)d9 ns2; since in d9 s2 configuration the d orbitals are one electron away from being completely filled, an electron from the s orbital occupies a d orbital instead, leaving one electron in t
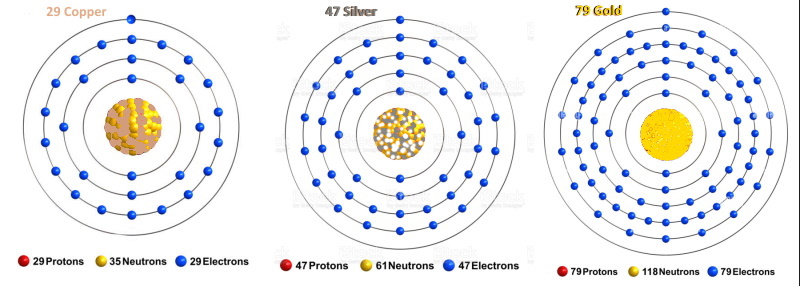
he valence shell. These metals usually form 1+ charges, which is why this group has historically been called 1B. The elements in this group are sometimes referred to as "coinage metals" because they have historically been used in coins, although other metals besides the ones in Group 1B have been used in coins as well. Copper (Cu, Z=29)Copper is a malleable, ductile, reddish-brown metal. Its name is derived from the Old English name coper, which is in turn derived from the Latin word cuprum for "from the island of Cyprus," which was the leading supplier of copper in the Mediterranean area at the time of the Roman empire. The symbol "Cu" is derived from cuprum. Copper is not an extremely common element in the Earth's crust, having a concentration of only 50 ppm in the Earth's crust (making it the 26th most abundant element), but it is relatively easy to obtain because it is highly concentrated in its ores. Some copper is found as the pure metal form in 99% or better purity; the ores of copper include chalcopyrite [CuFeS2], chalcocite [copper(I) sulfide, Cu2S], covellite [copper(II) sulfide, CuS], bornite [Cu5FeS4], cuprite [copper(I) oxide, Cu2O], tenorite [copper(II) oxide, CuO], paramelaconite, chalcanthite [copper(II) sulfate pentahydrate, CuSO4·5H2O], and and brochantite, malachite [copper(II) carbonate hydroxide, Cu2CO3(OH)2],and azurite [/a].
Copper artifacts have been found which date back to about 10,000 years ago; the refining of copper ores started at around 5000 BC. However, copper is a soft metal, and is not unsuitable for making tools and weapons. The Bronze Age began in around 3000 BC when it was discovered that copper could be alloyed with tin (at a ratio of about two parts copper to one part tin) to make bronze; the resulting alloy was much stronger, and was capable of holding an edge.
Copper is a good conductor of heat and electricity, and is used extensively in electrical wiring. For this usage, it must be extremely pure, because trace amounts of other metal decrease the electrical conductivity and increase the amount of resistance in copper wiring. Copper can be roasted and smelted from its ore in about 99% purity, but this is still not good enough for use as wiring. Copper is further refined in an electrochemical process, in which impure copper is used at the anode of the electrolytic cell, and a sheet of very pure copper is used as the cathode. Copper is oxidized at the anode into Cu2+ ions, which dissolved in the electrolyte solution, and are are reduced at the cathode, plating out on the electrode as copper metal in better than 99.6% purity. Other easily oxidized metal ions, such as Zn2+ and Fe2+ remain in the electrolyte solution, and less easily oxidized metals fall to the bottom of the cell as "anode mud," which contains such “impurities” as silver, gold, and platinum.
Copper was widely used to make cookware such as pans and kettles, but this use is less common now because of widely available cheaper cookware made of aluminum or stainless steel. Many high-quality pots and pans are made with copper bottoms, which distributes the heat from the stovetop quickly and evenly throughout the pan.
Because copper resists corrosion by water, air, and most acids (except for concentrated nitric and sulfuric acids), it is ideal for making coins. The US one-cent piece, the penny, used to be made from solid copper (or copper alloyed with tin, nickel, or zinc) but due to the increasing cost of copper, this was replaced in 1982 with a coin made from a zinc core surrounded by a thin copper plating (about 2.4% of the mass of the coin). [An interesting chemical demonstration can be done with these kinds of pennies: if the copper on the edge of the penny is filed off to expose the zinc, and the coin dropped into hydrochloric acid, the zinc will undergo a single-displacement reaction with the hydrochloric acid and dissolve in the solution as soluble zinc chloride, ZnCl2, while the copper on the obverse and reverse sides of the penny will remain unaffected, leaving two thin pieces of copper foil still bearing the impressions of the Lincoln head and the Lincoln Memorial.] The US five-cent piece, the nickel, is an alloy of 75% copper and 25% nickel; and the the ten-cent piece (the dime) and 25-cent piece (the quarter) are 91.67% copper and 8.33% nickel. The US Sacajawea dollar coin, first issued in November of 1999 is also made primarily of copper (88.5% copper, 6% zinc, 3.5% manganese, and 2% nickel).
Copper is also widely used in plumbing and water pipelines, motors and generators, circuitry and computer chips, household fixtures, kitchen utensils, dinnerware (such as sterling silver, which consists of silver alloyed with 7.5% copper or some other metal), in ceramic glazes and colored glass, and in musical instruments (especially those made of brass).
Copper is also widely used in statuary. The Statue of Liberty is made of 81.3 tons of copper plating mounted on a steel skeleton. The Colossus of Rhodes, one of the Seven Wonders of the Ancient World, was a 110-foot bronze statue of the Greek god Helios on the island of Rhodes which stood at the mouth of the Mandraki harbor entrance; it was completed in 280 BC and destroyed by an earthquake in 224 BC.
Copper is used to make several important metal alloys. Bronze is an alloy of copper and tin. Brass is an alloy of copper and zinc, which is addition to being harder than copper can be polished to a high, golden luster. Gunmetal (also known as red brass) is an alloy of copper, tin, and zinc which is strong enough to make guns and cannons. Cupronickel (also called Monel metal) is an alloy of copper and nickel commonly used in coins, such as the US 5-cent piece, and in shipbuilding. Alloys of copper and nickel are also used in desalination plants and underwater pumps because it resists corrosion by sea water. Aluminum bronzes are alloys of copper and up to 7% aluminum that have a gold luster, and are very resistant to corrosion; they are used in naval architecture, engine parts on ships, and landing gear components.
The most common oxidation states of iron are Cu+, the copper(I) or cuprous ion, and Cu2+, the copper(II) or cupric ion. Copper(II) sulfate, CuSO4, is white in is anhyrous form, and deep blue when it is complexed with water in the pentahydrate form, CuSO4·5H2O. This material, sometimes called blue vitrol, is used in fungicides and algicides, and in ink pigments. Copper(II) chloride, CuCl2, is used as a dye fixer in the textile industry. Copper(II) acetate, Cu(C2H3O2)2, also known as verdigris, forms as copper is exposed to air and seawater over long periods of time; it is widely used as a green pigment in oil paintings.
Copper is essential in the diet because it is a part of several enzymes in the body, such as cyctochrome c oxidase, which is required for energy production. Copper is plentiful in foods such as seafood (especially shellfish), lamb, duck, pork, and beef, almonds, walnuts, Brazil nuts, sunflower seeds, mushrooms, and bran. The minimum amount of copper needed in the diet is around 1.2 mg per day, but a typical diet can provide as much as 6 mg per day.
Some mollusks (such as oysters) and some arthropods (such as the horseshoe crab) use hemocyanin to carry oxygen to their cells. In this protein complex, a copper(I) ion is held in place by histidine groups; molecular oxygen complexes with the copper ion to form a blue copper(II)-oxygen complex. (In Star Trek, Vulcan blood was green because it was copper-based.)
Silver (Ag, Z=47)Silver is a soft, ductile, malleable, silvery (duh) metal. The name is derived from the Anglo-Saxon word for the metal, siolfur; its chemical symbol, Ag, is derived from the Latin name for silver, argentum. Silver is a fairly rare element, having a concentration of 70 ppb in the Earth's crust, making it the 66th most abundant element. It is found in the ores argentite and acanthite [both silver sulfide, Ag2S], stephanite [silver antimony sulfide, Ag5SbS4], horn silver [silver chloride, AgCl], silver arsenide [Ag3As], bromyrite [AgBr], and cerargyrite [AgCl]. However, most silver is obtained as a by-product from the refining of other metals.
Silver was known in many ancient civilizations, but since is not obtained from the ground as the pure metal, it has not been used for as long as gold has been. Silver was first mined around 3000 BC, and was extensively used in coins, although silver coins are too soft to be very durable.
Silver is the metal which is most conductive to electricity and heat, which makes is extremely important in the electronics industry. The metal is extremely malleable and ductile — one gram of silver can be drawn into a wire nearly two kilometers long. Silver is is stable to water and oxygen, but forms silver sulfide, Ag2S, when it is exposed to sulfur compounds in the air; this forms a black coating (tarnish) on the silver, which requires regular cleaning. Silver is considered one of the precious metals because of its rarity, stability, high melting point, and high luster.
Silver is widely used in the electronics industry in switches, circuits, and electronic devices (such as computer keyboards). It is also used in cutlery, jewelry, and mirrors. Sterling silver is an alloy of 93% silver and 7% copper which is widely used in tableware. Amalgams of silver, tin, and mercury are used by dentists to fill cavities.
In its compounds, silver typically is in the +1 oxidation state, but some +2 and +3 compounds are also known. Silver nitrate, AgNO3, is used in photographic emulsions, as are silver bromide, AgBr, silver chloride, AgCl, and silver iodide, AgI. Silver iodide is used in used to seed clouds to encourage rainfall.
Colloidal silver is a suspension of silver used in "alternative" medicine as a antibiotic. However, prolonged use, or overdoses, of colloidal silver can lead to a condition called argyria, in which silver becomes deposited in tissues throughout the body, causing the skin to become bluish-gray. Although this is not harmful in itself, it is disfiguring, and it may not be possible to reverse the condition.
Gold (Au, Z=79)Gold is a soft, malleable, yellow metal. It is an extremely rare element, having a concentration of 1 ppb in the Earth's crust, making it the 73rd most abundant element. The name is derived from the Anglo-Saxon language, while the chemical symbol "Au" is derived from the Latin name for the metal, aurum ("shining dawn"). It is often found as the free element as nuggets or grains, and in alluvial deposits, and is associated with some sulfide ores, and is also found in the mineral sylvanite [silver gold telluride, (Ag,Au)Te2].
Gold has been used by humans for thousands of years. Since uncombined gold can be obtained directly from the ground (or by panning streams and rivers), and is easy to refine because it has a relatively low melting point (1064ºC) it was one of the first metals to be discovered, and was widely used in making jewelry, coins, and other artifacts. The desire for gold encouraged the ancient alchemists to try to figure out how to make the "Philosopher's Stone" that would enable them to transmute base metals such as lead into gold. Although they never succeeded (except perhaps for Nicholas Flamel, in the Harry Potter books by J. K. Rowling), they did accumulate a lot of empirical observations that led to the development of the science of chemistry.
Gold is extremely unreactive: the only acids that dissolve gold are aqua regia ("king of waters," a mixture of concentrated hydrochloric acid and nitric acid), and selenic acid, H2SeO4. Gold extremely malleable and ductile: one gram of gold can be beaten out to make a thin film that is one square meter in area and 50 nanometers in thickness. Gold is also a very good electrical conductor, making it useful in electrical connections. The oceans contain roughly 10 million tons of gold, but it is at such a low concentration — about 10 parts per thousand — that is impractical to try to extract it. Like silver, gold is considered one of the precious metals because of its rarity and stability.
Gold is used extensively in jewelry (this accounts for about 75% of all gold produced), in bullion (precious metals in bulk form), and in electronics. Thin films of gold are used in some large buildings to reflect away heat; the Mylar film that coats the skins of some spacecraft is also covered in gold foil for the same reason.
The purity of gold is measured in units called karats. Pure gold is 24 karat. An alloy that is 92% gold is 22 carat (92% of 24), 18 carat gold is 75% gold, 12 karat is 50% gold, and so on. Gold in jewelry is often alloyed with silver or copper, with a small amount of zinc to harden it. Gold can be given a range of colors depending on the metal with which it is alloyed: white gold contains 10% nickel, red gold contains 50% copper, blue gold contains 54% indium, purple gold contains 20% aluminum, green gold (also known as electrum) contains 27% silver, and black gold contains 25% cobalt.
In its compounds, gold is usually found in the +1 or +3 oxidation states; Au+ is known as the gold(I) or aurous ion, and Au3+ is known as the gold(III) or auric ion.
Gold salts are used in some treatments for arthritis, when non-steroidal anti-inflammatory drugs do not work. Because there are side effects from having gold build up in the body, this kind of treatment can only be used for a few years. Gold is also used in dentistry to fill cavities and make crowns; the gold is alloyed with silver, palladium, and zinc to harden the amalgam. Over 60 tons of gold per year are used in this fashion.
[/a]