jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 4:57:53 GMT -5
Had a double header yesterday. a batch out of the vibe and a batch out of the kiln. Start with the kiln. These were heated twice. First time shards covered with protective layer of clear sheet glass or sawn from a melt brick or melted colored chunks. Small kiln packed to the hilt with 8 inch ceramic shelf tiles. The best ones are shards from the glass blower, the others are my assemblies. About all useless to a glass fuser because they do not have a flame polish so they get a trip to the tumbler for their needed polish. A few need a third trip to the kiln. All are minimally ground or shaped before and after both melts. Lots of work.   These came out of the vibe. All have flat backs   These 2 look fine from the front but the backside had some foreign yellow glass that was not melt compatible.  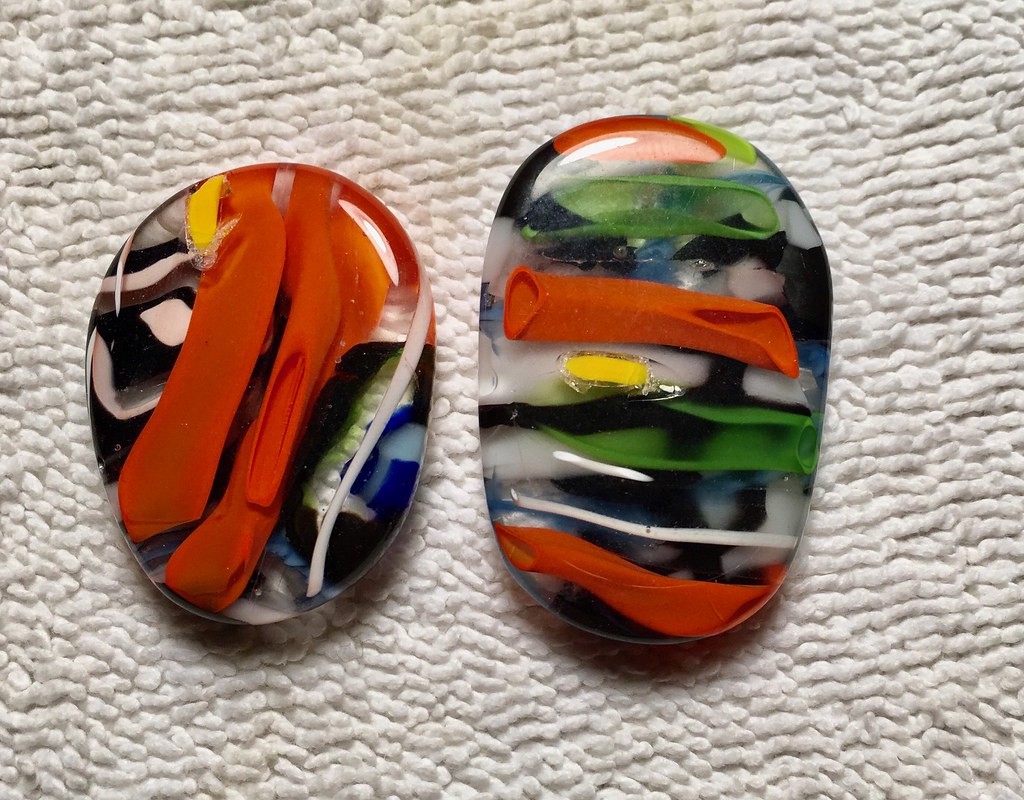 Textures    Fritted and frit streaked   bright  Misc.     Chinese junk bead melt  Sawn from melt brick - confetti  And thanks for looking. |
|
|
|
Post by fernwood on Mar 18, 2018 5:01:53 GMT -5
Mesmerizing to look at all the different textures.
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 5:23:30 GMT -5
Mesmerizing to look at all the different textures. Slowly dialing in these melts fernwood. It is so complicated for a newbie. Melting larger than needed and then grinding down to desired size for final remelt is working best. So much raw glass in so many forms requires about 6 to 8 different methods every time at first melt. The grind to size/shape after re-fire is easy. It is the first melt that creates the challenge due to volume, placement, color location and mix, color coordination, etc. Figuring lose of size/shape due to tumbling an added responsibility. Lol, the pre-composed shards from the glass blower are easiest meaning that I suck at composition which does not bother me in the least. Just trying to produce pretty pendants. I did go out and purchase frits and stringers to assist composition instead of making/using my own frit. |
|
|
|
Post by vegasjames on Mar 18, 2018 5:35:17 GMT -5
Very cool looking.
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 5:39:16 GMT -5
The younger glass blower called yesterday and asked me to come get his scrap. He had classes on heavy fritting. Got quite a bit of this glass loaded with a thick color layer. Must extract and melt to pendants. Extraction involves grinding away most of the clear glass before melting to avoid colors leaching out into excess clear glass. Probably not an issue with this heavy color layer.  |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 5:49:31 GMT -5
I really want to get into some reactive agents James. The earthy textures they produce should go over well. Wishing I had your chemical mind. If you know of any metal powders or chemical derivatives from rocks I would sure appreciate it. One of the stores in town sells a giant plate slumped in Greece that has a smattering of various reactive agents and pieces of metal and screen mixed in. Funny, a lot of the glass blowers no longer make their own grit and colored glass. I see why after crushing out some colors of frit and screening it to size. Lots of work. I did learn to heat colored glass to 600F in a convection oven and chunk it in ice water to assist the crushing process. That certainly gives the ram rod operator an upper hand. Kinda feels good to cripple that hard crap with hundreds of shock fractures. |
|
|
|
Post by vegasjames on Mar 18, 2018 6:39:46 GMT -5
I really want to get into some reactive agents James. The earthy textures they produce should go over well. Wishing I had your chemical mind. If you know of any metal powders or chemical derivatives from rocks I would sure appreciate it. One of the stores in town sells a giant plate slumped in Greece that has a smattering of various reactive agents and pieces of metal and screen mixed in. Funny, a lot of the glass blowers no longer make their own grit and colored glass. I see why after crushing out some colors of frit and screening it to size. Lots of work. I did learn to heat colored glass to 600F in a convection oven and chunk it in ice water to assist the crushing process. That certainly gives the ram rod operator an upper hand. Kinda feels good to cripple that hard crap with hundreds of shock fractures. It would help if I knew exactly what you are trying to achieve. Copper can be reactive in glass. If you melt copper in to the glass then expose areas of the glass to UV light. Then the glass is annealed which "develops" the copper where exposed to the UV light. Can make for some interesting effects. I know bubbles are induced in decorative glass usually using silica or I think it was tin places between the pieces of glass they wanted bubbles. There are additives that can be used to increase RI, hardness, opacity, etc. Most though are going to be in complete melts, not just slumping. So again if you can be more specific I can see if there is anything I can tell you. Could be interesting to try some of the glass in an reduction atmosphere as well to see what you end up with. |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 6:58:59 GMT -5
I really want to get into some reactive agents James. The earthy textures they produce should go over well. Wishing I had your chemical mind. If you know of any metal powders or chemical derivatives from rocks I would sure appreciate it. One of the stores in town sells a giant plate slumped in Greece that has a smattering of various reactive agents and pieces of metal and screen mixed in. Funny, a lot of the glass blowers no longer make their own grit and colored glass. I see why after crushing out some colors of frit and screening it to size. Lots of work. I did learn to heat colored glass to 600F in a convection oven and chunk it in ice water to assist the crushing process. That certainly gives the ram rod operator an upper hand. Kinda feels good to cripple that hard crap with hundreds of shock fractures. It would help if I knew exactly what you are trying to achieve. Copper can be reactive in glass. If you melt copper in to the glass then expose areas of the glass to UV light. Then the glass is annealed which "develops" the copper where exposed to the UV light. Can make for some interesting effects. I know bubbles are induced in decorative glass usually using silica or I think it was tin places between the pieces of glass they wanted bubbles. There are additives that can be used to increase RI, hardness, opacity, etc. Most though are going to be in complete melts, not just slumping. So again if you can be more specific I can see if there is anything I can tell you. Could be interesting to try some of the glass in an reduction atmosphere as well to see what you end up with. Thanks James, this is all greek to me. I get the bubbles from silica, tin and copper and all only to a degree. Being that I take 100+ small pieces of glass to ~1500F there is always room for test pieces. I get what you are saying about 1500F being low for many chemical inputs. Always have a clear piece of glass to throw some trash on if anything comes to mind as long as it doesn't explode too violently. Throw it at me if you feel so inclined. |
|
|
|
Post by MrP on Mar 18, 2018 7:32:29 GMT -5
James I am wondering if you have found any glass that will not take a FINE polish. My wife really likes glass so I have bought a lot of glass chunks and have found a few that just will not get a brilliant shine. On some pieces I can see micro pores, not really bubble holes but just ultra fine texture. I should say that overall the pieces have a great shine but if looked at close the micro pores do show...................MrP
|
|
|
|
Post by fernwood on Mar 18, 2018 9:21:58 GMT -5
I really want to get into some reactive agents James. The earthy textures they produce should go over well. Wishing I had your chemical mind. If you know of any metal powders or chemical derivatives from rocks I would sure appreciate it. One of the stores in town sells a giant plate slumped in Greece that has a smattering of various reactive agents and pieces of metal and screen mixed in. Funny, a lot of the glass blowers no longer make their own grit and colored glass. I see why after crushing out some colors of frit and screening it to size. Lots of work. I did learn to heat colored glass to 600F in a convection oven and chunk it in ice water to assist the crushing process. That certainly gives the ram rod operator an upper hand. Kinda feels good to cripple that hard crap with hundreds of shock fractures. I am by no means a chemist, but have some thoughts. Have you considered placing small rock chips between 2 layers of glass and just see what happens? I know quite a few people who would love something like this. I have been crushing some crystal rocks like Citrine, Amethyst, Peridot and Rose Quartz. Then placing them in little glass bottles. Also did Turquoise and Specular Hematite. They are being used as pendulums, necklaces and sun catchers. Now am doing custom orders this way with specific stones. Some stones might not survive the heat, but others should. I try to select chips with great color and shapes most of the time. The leftover powder is used for Medicine Wheels. Just a thought of something new to try. |
|
|
|
Post by orrum on Mar 18, 2018 9:35:42 GMT -5
Looks great Jim! I seriously need a kiln...
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 9:58:07 GMT -5
James I am wondering if you have found any glass that will not take a FINE polish. My wife really likes glass so I have bought a lot of glass chunks and have found a few that just will not get a brilliant shine. On some pieces I can see micro pores, not really bubble holes but just ultra fine texture. I should say that overall the pieces have a great shine but if looked at close the micro pores do show...................MrP Yep, glass varies in hardness and amount of those tiny micro pores. Same with obsidian, mahogany is one of the harder obsidians. When using the nippers to break glass into pieces some nips easily and some will test your grip. I work with soda glass mostly and it is considered soft glass. If it nips easy then it usually does not polish as well. But sort glass can have intense stress in it which can make it hard. One harder glass would be test tubes and scientific chemical containers like flasks. Easy to polish. I believe they are Pyrex or borosilicate glass with a low COE of 33 so they can be heated without expanding and shrinking with temperature. Apparently glass can be boiled and that may cause the most bubbles and micro pores. some impurities causes bubbles. The soft glass I post here starts to boil 100 to 200 degrees higher than I heat at 1500. So 1600 to 1700F to boil. The bubbles start at the bottom I believe. and can transport colored glass as they pass thru a colored glass layer. Above my skill level. Micro pores - so what, most need magnification and are not seen by average person. |
|
|
|
Post by MrP on Mar 18, 2018 10:32:32 GMT -5
James I am wondering if you have found any glass that will not take a FINE polish. My wife really likes glass so I have bought a lot of glass chunks and have found a few that just will not get a brilliant shine. On some pieces I can see micro pores, not really bubble holes but just ultra fine texture. I should say that overall the pieces have a great shine but if looked at close the micro pores do show...................MrP Yep, glass varies in hardness and amount of those tiny micro pores. Same with obsidian, mahogany is one of the harder obsidians. When using the nippers to break glass into pieces some nips easily and some will test your grip. I work with soda glass mostly and it is considered soft glass. If it nips easy then it usually does not polish as well. But sort glass can have intense stress in it which can make it hard. One harder glass would be test tubes and scientific chemical containers like flasks. Easy to polish. I believe they are Pyrex or borosilicate glass with a low COE of 33 so they can be heated without expanding and shrinking with temperature. Apparently glass can be boiled and that may cause the most bubbles and micro pores. some impurities causes bubbles. The soft glass I post here starts to boil 100 to 200 degrees higher than I heat at 1500. So 1600 to 1700F to boil. The bubbles start at the bottom I believe. and can transport colored glass as they pass thru a colored glass layer. Above my skill level. Micro pores - so what, most need magnification and are not seen by average person. Yes James I know that but once I see it I can't 'unsee' it, and then it looks bigger then life! I have noticed that it takes very little to polish some glass. When I pre-shape pendants I can tell there is a great difference in the different glass. I have a block of light yellow glass that is as heavy as lead, cuts like butter but factors easily when cut as a slab..........................MrP |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 10:33:09 GMT -5
I really want to get into some reactive agents James. The earthy textures they produce should go over well. Wishing I had your chemical mind. If you know of any metal powders or chemical derivatives from rocks I would sure appreciate it. One of the stores in town sells a giant plate slumped in Greece that has a smattering of various reactive agents and pieces of metal and screen mixed in. Funny, a lot of the glass blowers no longer make their own grit and colored glass. I see why after crushing out some colors of frit and screening it to size. Lots of work. I did learn to heat colored glass to 600F in a convection oven and chunk it in ice water to assist the crushing process. That certainly gives the ram rod operator an upper hand. Kinda feels good to cripple that hard crap with hundreds of shock fractures. I am by no means a chemist, but have some thoughts. Have you considered placing small rock chips between 2 layers of glass and just see what happens? I know quite a few people who would love something like this. I have been crushing some crystal rocks like Citrine, Amethyst, Peridot and Rose Quartz. Then placing them in little glass bottles. Also did Turquoise and Specular Hematite. They are being used as pendulums, necklaces and sun catchers. Now am doing custom orders this way with specific stones. Some stones might not survive the heat, but others should. I try to select chips with great color and shapes most of the time. The leftover powder is used for Medicine Wheels. Just a thought of something new to try. I have heat treated agate and coral and it usually shatters. Garnets are supposed to be one of the best at taking heat. Hematite and turquoise sound like great candidates fernwood. Powdered forms, are they easier to melt in ? |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 10:35:57 GMT -5
Looks great Jim! I seriously need a kiln... I bet you could melt those toilets you collect into fine jewels lol. Still laugh about all your pretty rocks in the back of the truck with a broken toilet thrown in. Perplexing those out west no doubt. |
|
|
|
Post by rockjunquie on Mar 18, 2018 10:41:55 GMT -5
Looking great!!!!! Love all that color!
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 10:44:38 GMT -5
Look past such MrP. If such a defect is hard to see 18 inches away then be done with it. Pretty sure this soft glass I work with is no doubt heavy. Limestone is heavy and is a major ingredient in glass. There is some obvious ingredient that makes it denser, got to be. The COE glass I have is 90 96 and 104. By far mostly 96 as that is what the scrap glass is. Borosilicate glass is COE 33, I will look up it's density and compare it to soft glass. Don't know if old lead glass or gold glass is heavier, I don't think they use much lead or gold when they add it. composition of glass gets complicated |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 10:45:56 GMT -5
Looking great!!!!! Love all that color! Tela likes color, I know that. Me too Tela. It does something to the endorphins. |
|
|
|
Post by hummingbirdstones on Mar 18, 2018 13:33:41 GMT -5
Looking good, jamesp! Those heavy frit pieces are yummy. |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,155
|
Post by jamesp on Mar 18, 2018 14:43:05 GMT -5
Looking good, jamesp! Those heavy frit pieces are yummy. When you walk in this glass blower's shop he has a bulletin board with about 50 different frit packs in clear baggies so you can pick your color. Apparently has a whole bunch more in proprietary storage. I was just prepping some for melt. when the frontal chunk is ground flat and the piece is wet that color is hard to describe with a camera. Not to mention the flowing effect. It's a trip and am anxious to get some of them polished. I am going to contract a rough globe of that frit pattern blown to make pendants from. Can't control myself. |
|