|
|
Post by 1dave on Mar 3, 2018 12:43:54 GMT -5
realsalt.com/ Me: "So real salt contains sodium, but also magnesium, potassium, silver, gold, diamonds, rubies . . . Wife: No, not diamonds and rubies! Me: Oh. So this is the cheap variety.
|
|
|
|
Post by MsAli on Mar 3, 2018 15:49:55 GMT -5
I always use the sea salt.
Pink and black are my favorite
You don't need as much to get the flavour
|
|
|
|
Post by grumpybill on Mar 3, 2018 17:07:22 GMT -5
I'm on a sodium restricted diet. I don't remember what salt ("real" or otherwise) tastes like.
|
|
|
|
Post by vegasjames on Mar 3, 2018 17:20:37 GMT -5
I rarely use salt on my foods, but when I do I prefer the Real (Himalayan) Salt. It does provide more flavor. The salt is mined from ancient sea beds and is the same salt used to make the salt lamps. So during margarita parties you can just lick the lamp for the salt.  |
|
|
|
Post by MsAli on Mar 3, 2018 19:25:36 GMT -5
I rarely use salt on my foods, but when I do I prefer the Real (Himalayan) Salt. It does provide more flavor. The salt is mined from ancient sea beds and is the same salt used to make the salt lamps. So during margarita parties you can just lick the lamp for the salt.  I've licked my lamp...don't recommend it |
|
|
|
Post by vegasjames on Mar 3, 2018 19:52:22 GMT -5
I rarely use salt on my foods, but when I do I prefer the Real (Himalayan) Salt. It does provide more flavor. The salt is mined from ancient sea beds and is the same salt used to make the salt lamps. So during margarita parties you can just lick the lamp for the salt.  I've licked my lamp...don't recommend it I f the person is drunk enough already then I doubt they would care.  |
|
|
|
Post by MsAli on Mar 3, 2018 21:14:52 GMT -5
I've licked my lamp...don't recommend it I f the person is drunk enough already then I doubt they would care.  Sad thing is I wasn't drunk....just wanted to know what it tasted like  |
|
|
|
Post by vegasjames on Mar 3, 2018 21:15:59 GMT -5
I f the person is drunk enough already then I doubt they would care.  Sad thing is I wasn't drunk....just wanted to know what it tasted like  At least you did not lick the outlet that it plugs in to.  |
|
|
|
Post by MsAli on Mar 3, 2018 21:18:44 GMT -5
Sad thing is I wasn't drunk....just wanted to know what it tasted like  At least you did not lick the outlet that it plugs in to.  I'm not that dumb  |
|
|
|
Post by fantastic5 on Mar 3, 2018 21:47:18 GMT -5
At least you did not lick the outlet that it plugs in to.  I'm not that dumb  I licked my salt lamp too! I thought it tasted like nice Himalaya salt. But I LOVE the taste of salt, always have. As a kid growing up in Michigan I used to eat the road salt on my way to elementary school. On second thought, maybe I shouldn't admit to that. Lol |
|
|
|
Post by MsAli on Mar 3, 2018 21:53:25 GMT -5
I'm not that dumb  I licked my salt lamp too! I thought it tasted like nice Himalaya salt. But I LOVE the taste of salt, always have. As a kid growing up in Michigan I used to eat the road salt on my way to elementary school. On second thought, maybe I shouldn't admit to that. Lol  Ok I've never done that!! I did stick my tongue to a really pretty sparkly icy metal pole once though |
|
|
|
Post by vegasjames on Mar 4, 2018 5:57:14 GMT -5
I'm not that dumb  I licked my salt lamp too! I thought it tasted like nice Himalaya salt. But I LOVE the taste of salt, always have. As a kid growing up in Michigan I used to eat the road salt on my way to elementary school. On second thought, maybe I shouldn't admit to that. Lol A little lead from the car exhaust back then never hurt anyone.  |
|
|
|
Post by kk on Mar 4, 2018 6:45:52 GMT -5
And yet, even "Real" salt comes with an expiry date.    Funny enough, I was just talking about salt to my hiking partner today, and neither of us could come up with an explanation as to how salt came to be. Considering that salt is pretty much in everything on this planet, we concluded that, just like water, there is no change in quantity of this substance on the planet. But then the question was raised as to how it formed.  ? |
|
|
|
Post by fernwood on Mar 4, 2018 7:22:51 GMT -5
I prefer the taste of Himalayan salt. Plus, the process to turn it into lamps does add some preservatives which one should not eat. The Himalayan has minerals that our bodies need.
Another great way to get pure salt, is to evaporate ocean water, from a source with no contaminants. Know everything that is in the local where you are harvesting the water form. Best option is to travel out, via boat, at least 200 yards from shore, sometimes more, depending on the water depth. Lower a collection vessel down about 50 yards, collect the water. Then test or have it tested for impurities. If it is free from these, then go back with a larger collection vessel and collect a bunch. If not free, then try another sample at a greater depth.
The natural evaporation process will take a while. This can be sped up, if you have the resources and time. If you need specific supplements, consider adding those to the sea water prior to evaporation. Mix well. Some might need the sea water to be heated for full blending.
another option, readily available, are the salt domes in the USA. Some are off limits to the public now, due to storage of nuclear waste, but others are waiting to be found. Depending on location, these may contain many health beneficial minerals as well.
JMHO
|
|
|
|
Post by 1dave on Mar 4, 2018 9:44:40 GMT -5
And yet, even "Real" salt comes with an expiry date.    Funny enough, I was just talking about salt to my hiking partner today, and neither of us could come up with an explanation as to how salt came to be. Considering that salt is pretty much in everything on this planet, we concluded that, just like water, there is no change in quantity of this substance on the planet. But then the question was raised as to how it formed.  ? Of all the blobs of matter that collected to form Earth, the lightest and most common have remained to form the crust. The 8 most common in order are: oxygen, silicon, aluminum, iron, calcium, sodium potassium, and magnesium. The oceans are a different matter. The halogens (salt producing) are a group in the periodic table consisting of five chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). When halogens react with metals they produce a wide range of salts, including calcium fluoride, sodium chloride (common table salt), silver bromide and potassium iodide. In the modern IUPAC nomenclature, this group is known as group 17. The symbol X is often used generically to refer to any halogen.  Because of its great reactivity, all chlorine in the Earth's crust is in the form of ionic chloride compounds, which includes table salt. It is the second-most abundant halogen (after fluorine) and twenty-first most abundant chemical element in Earth's crust. These crustal deposits are nevertheless dwarfed by the huge reserves of chloride in seawater. Although the sodium and Chlorine are separate ions, the attraction between the Na+ and Cl− ions is so strong that they easily join to form compoinds. In the solid state the bond is so strong that only highly polar solvents like water can dissolve NaCl well. The pH of a sodium chloride solution remains ≈7 due to the extremely weak basicity of the Cl− ion, which is the conjugate base of the strong acid HCl. In other words, NaCl has no effect on system pH. As continental plates ping-pong from super continent to super continent, oceans are formed and dried out to form salt beds consisting mostly of sodium chloride.  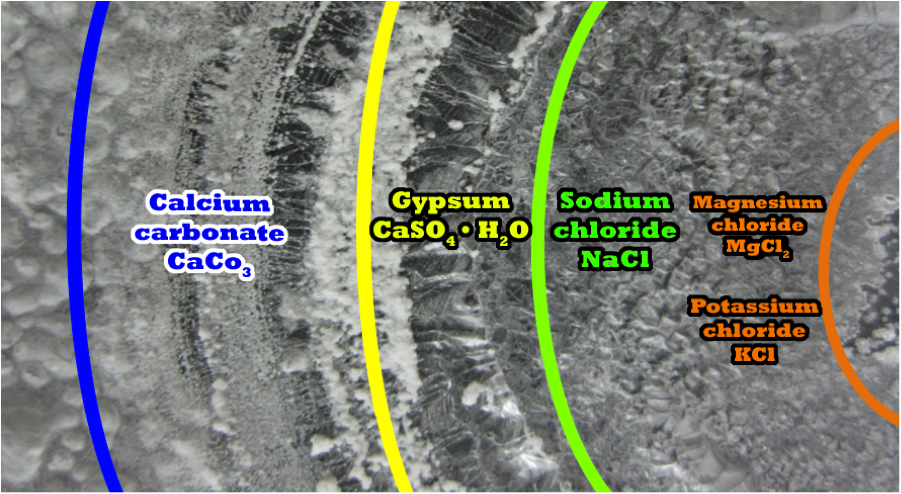 SeaWater Salt Rings Formed on a Watch Glass Outer ring, least soluble salt, precipitated first; inner ring, most soluble salt, precipitated last. Read more: forum.rocktumblinghobby.com/thread/81933/rocks-belong#ixzz58n8G63d8 |
|
|
|
Post by kk on Mar 4, 2018 9:59:12 GMT -5
So that means while there is a "salt-circle", our consumption is small enough to avoid a finite termination point for salt?
|
|
|
|
Post by 1dave on Mar 4, 2018 10:04:08 GMT -5
|
|
|
|
Post by MsAli on Mar 4, 2018 10:42:44 GMT -5
Interesting article
I have severe leg cramps at night and started drinking Lifewater or Smartwater and it has made a huge difference.
Avoid Gatorade and other high sugar drinks with electrolytes.
|
|
|
|
Post by grumpybill on Mar 4, 2018 13:00:51 GMT -5
Older adults with decreased kidney function...<clip>...face a heightened risk of hyponatremia. Interesting. I'm an "older adult with decreased kidney function" and my nephrologist wants me to severely restrict my sodium intake in an effort to prolong the useful life of my kidneys. |
|
|
|
Post by 1dave on Mar 4, 2018 14:55:00 GMT -5
Interesting article I have severe leg cramps at night and started drinking Lifewater or Smartwater and it has made a huge difference. Avoid Gatorade and other high sugar drinks with electrolytes. Me too! as soon as they start I stand up and take a magnesium pill. walk around for a bit and they go away. |
|