herb
spending too much on rocks
 
Member since November 2011
Posts: 445
|
Post by herb on Jan 15, 2024 11:04:21 GMT -5
Knowing nothing about glass fusing, I am curious if the piece with the fractures couldn't be saved? Could you not reheat it slowly to the point where the glass melts enough to fuse together all the fractures? Or can you not get the glass fluid enough for the fractures to properly heal? Maybe the air in the fractures cant "bubble" out so there will still be defects? Inquiring minds what to know! To save energy two bowl/brick melts were performed simultaneously. Peak temp(1475F) dwell time was raised from 3 hours to 4 hours to assure kiln would not struggle to melt 25 pounds total glass. The above brick being brown and black glass could handle the extra heat. However the red glass in the other brick suffered from too much heat and fractured trashing 11 pounds of glass grrr. At raw glass cost of about $15/pound it is good that I acquired this glass very cheap.  Has cracks throughout(trash):  |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 16, 2024 15:33:32 GMT -5
Knowing nothing about glass fusing, I am curious if the piece with the fractures couldn't be saved? Could you not reheat it slowly to the point where the glass melts enough to fuse together all the fractures? Or can you not get the glass fluid enough for the fractures to properly heal? Maybe the air in the fractures cant "bubble" out so there will still be defects? Inquiring minds what to know! To save energy two bowl/brick melts were performed simultaneously. Peak temp(1475F) dwell time was raised from 3 hours to 4 hours to assure kiln would not struggle to melt 25 pounds total glass. The above brick being brown and black glass could handle the extra heat. However the red glass in the other brick suffered from too much heat and fractured trashing 11 pounds of glass grrr. At raw glass cost of about $15/pound it is good that I acquired this glass very cheap.  Has cracks throughout(trash):  Herb this glass gets brittle when remelted. It casts one shot excellent. It is not a high quality fusing glass in that way, but it makes very strong glass that rarely fractures upon cooling in thick blocks. Analogy, large and powerful but not quick and nimble. Glass has many trade-offs too. Attempt to take obsidian to fuse/melt temps and most of it will convert to pumice, something in the composition makes it fizzle air bubbles intensely. |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 16, 2024 15:41:33 GMT -5
Another tribal pattern did as planned. A thinner arrowhead will reveal the translucent purple glass better than a 5/16" thick slab.  backlit 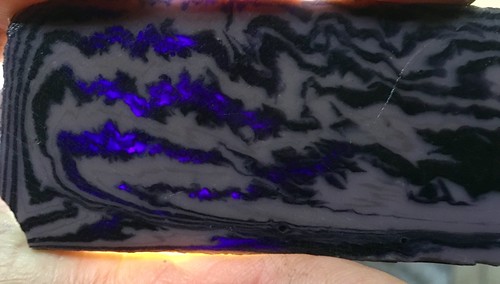 |
|
rockbrain
Cave Dweller 
Member since January 2022
Posts: 2,563
|
Post by rockbrain on Jan 16, 2024 15:59:28 GMT -5
That's sweet!
|
|
Fossilman
Cave Dweller 
Member since January 2009
Posts: 20,685 
|
Post by Fossilman on Jan 19, 2024 11:51:27 GMT -5
Doing a great job my friend!
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 19, 2024 12:26:51 GMT -5
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 19, 2024 12:27:34 GMT -5
Doing a great job my friend! Thanks fossilman. |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 19, 2024 12:44:55 GMT -5
Will be trying to repeat this 2018 pour. Used a smaller hope in the pour bowl and raised the bowl higher. Somehow the glass poured in thin stream that spiraled as it accumulated and piled in the mold, it also squashed outwardly. The complete dynamics remain a mystery.  Note how glass color changes after fusion:  Note how colors stayed in order down pontil pipe, but note spiral landing in mold:  slabs  slabs closer up, more complex up close:  tumbled but wet:  Trying to coax rockbrain to get a glass kiln... |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 19, 2024 12:53:48 GMT -5
This one should be a wild thang. Even poured it thru a smaller hole to increase mixing. In anneal mode till tonight.  |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 20, 2024 15:17:49 GMT -5
Melt above. Made a boo boo, forgot to put the 3/16" ceramic felt around the inside of brickmold. Well, glass expands when it solidifies, and it did just that requiring hammerized removal. NO other way to remove. Live/learn. Great tumbling fodder, oh well. Or cab fodder if anyone wants to give it a go for a cab just say. Used a bit too much dark glass. Brick, and the expansion sent fractures. Mold is strong/rigid/unbendable/etc:  Hammered results, quite psycho. get back fordite !:    Down the inverted volcano pour point:  misc chips  |
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 20, 2024 15:53:27 GMT -5
Will repeat above psych pour with following changes: 1) doubled the size of the pour hole to reduce mixing intensity 2) tripled/quadrupled the size of the glass chips to increase size of each color patch 3) went with brighter colors and avoided browns/blacks 13 pounds   |
|
|
|
Post by liveoak on Jan 21, 2024 7:41:47 GMT -5
Great tumbling fodder, oh well. Or cab fodder if anyone wants to give it a go for a cab just say. I'm game to try to cab a piece of "Jimite". Pretty funky looking.
Patty
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 21, 2024 9:20:00 GMT -5
Great tumbling fodder, oh well. Or cab fodder if anyone wants to give it a go for a cab just say. I'm game to try to cab a piece of "Jimite". Pretty funky looking.
Patty
Patent Pending on the "Jimite"... lol, sure thing, I'll get it to you Patty. |
|
|
|
Post by liveoak on Jan 21, 2024 9:24:36 GMT -5
Excellent Jim, I've been needing a day on the cabber- that would be a terrific excuse.
Credit goes to Tom on the "Jimite"
Just let me know the postage.
Patty
|
|
jamesp
Cave Dweller 
Member since October 2012
Posts: 36,169
|
Post by jamesp on Jan 24, 2024 15:14:17 GMT -5
Classic case of one glass color(the red in this case)trashing the whole project. There is some out-of-spec red glass in the batch no doubt. Irritating. see fractures when dry, it was the red that propagated all the fractures in the other colors:  wet showing colors better, cracks still visible. Just goes to show that conditions have to be favorable for obsidian to not crack, it can be varying temp or chemical issues. It is actually a miracle obsidian can be formed without controlled conditions.  |
|
|
|
Post by rockjunquie on Jan 24, 2024 16:14:34 GMT -5
Classic case of one glass color(the red in this case)trashing the whole project. There is some out-of-spec red glass in the batch no doubt. Irritating. see fractures when dry, it was the red that propagated all the fractures in the other colors:  wet showing colors better, cracks still visible. Just goes to show that conditions have to be favorable for obsidian to not crack, it can be varying temp or chemical issues. It is actually a miracle obsidian can be formed without controlled conditions.  Too bad. Those bottom ones are really nice. |
|
|
|
Post by liveoak on Jan 25, 2024 7:53:13 GMT -5
Can you remelt the failures ?
Hope so
Patty
|
|
|
|
Post by 1dave on Jan 25, 2024 15:33:04 GMT -5
jamesp Good looking glass bricks, even the ones that did not quite come out. Also good to see you active and posting again, your posts with pictures are always a favorite of mine. Stay busy my friend. Henry I agree HankRocks and jamesp |
|
|
|
Post by 1dave on Jan 25, 2024 15:46:39 GMT -5
I'll be interested to see what you make with this. I hope this works out well for you. Thank you much rockbrain. This was about the only affordable choice. Shoulda added lavender and purple to complete the natural spectrum. This comes in 7mm-10mm rods, could get cheaper if bought in plates or bricks. Consider the rods offer a whole different dimension in patterns. Certain I will be tooting my horn by posting photos  . Your toots are always so interesting! |
|
|
|
Post by 1dave on Jan 25, 2024 16:14:40 GMT -5
jamesp : Cobalt(II) chloride is an inorganic compound, a salt of cobalt and chlorine, with the formula CoCl2. The compound forms several hydrates CoCl2·nH2O, The anhydrous form is a blue crystalline solid; the dihydrate is purple and the hexahydrate is pink. Entrepreneurs have made many uses of this for weather prediction Items for sale. It works as a dye, and as a paint. I'd be interested in seeing how it works in glass. 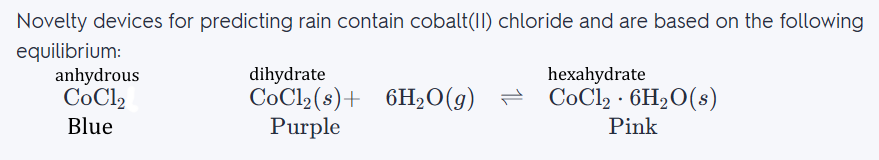  Dyed Cloth:  Painted owl:  Moisture Test Paper:  |
|